 We’re going to dive right in to listener questions in this Pinks and Blues episode because today is all about testing for Chlorine. Chlorine is the most well-known of the Halogen elements, a group of Oxidizing Agents. So, if you catch me using these words interchangeably, it’s because they are!
We’re going to dive right in to listener questions in this Pinks and Blues episode because today is all about testing for Chlorine. Chlorine is the most well-known of the Halogen elements, a group of Oxidizing Agents. So, if you catch me using these words interchangeably, it’s because they are!
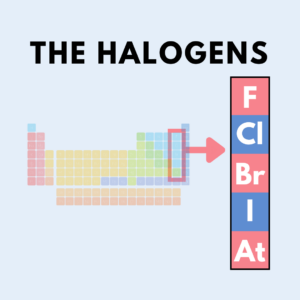
As a quick chemistry refresher, a Halogen is the chemical that forms a salt when it reacts with a metal. There are 5 Halogens in the Periodic Table: Fluorine, Chlorine, Bromine, Iodine, and Astatine and you can find the whole column of Halogens the column second furthest to the right.
In Cooling Towers, we typically use Chlorine or Bromine. By the way, whatever you use, make sure it is registered for that usage. There are two methods of testing Halogen: OT (Orthotolidine) and DPD (N, N-diethyl-p-phenylenediamine), which is the method many of us prefer because it allows us to have a free or total test. Are you curious yet about testing for Halogens?
Today, we are going to talk about all things Halogens. From differentiating the types of Halogens, to understanding the different testing methods, and to the importance of having a Water Management Plan to make our jobs easier.
Bottom line: Let’s learn about testing for Halogen in a Water System.
Your roadside friend, as you travel from client to client.
-Trace
Timestamps:
Starting a podcast, being a guest on other’s podcasts, accomplishing your goals [01:01]
Events in Water Treatment [09:54]
Thinking On Water With James [15:29]
Answering your questions for this week’s Pinks and Blues: Testing for Halogen in the System [16:39]
Identifying Halogens [19:01]
Understanding the different HalogenTests: OT Test (Orthotolidine) and the DPD Method (N, N-diethyl-p-phenylenediamine) [22:31]
Too much Halogen in the system [26:00]
Interferences with Halogen Tests [28:28]
Differentiating Free, Combined, and Total Halogen [32:03]
Identifying the best test to use [35:59]
The advantages of having a water management plan [39:09]
Put it into practice [42:51]
Thinking On Water With James:
In this week’s episode, we’re thinking about whether the brine tank on your softener system reaches 100% saturation between softener regenerations. Do you know? Have you ever measured % brine saturation in your brine? How would you measure % saturation? When is the best time to measure it? If it is not reaching 100% saturation, can anything be done to improve it? What is the impact upon softener regeneration with less than 100% brine saturation? Take this week to think about the % brine saturation in your softener system’s brine tank.
Quotes:
“A Halogen is a chemical that forms a salt when it reacts with a metal, and there are five Halogens in the Periodic Table: Fluorine, Chlorine, Bromine, Iodine, and Astatine.” – Trace Blackmore
“Halogens are highly reactive, which means they are quick to form bonds with other elements.” – Trace Blackmore
“Oxidizers are solids, liquids, or gasses that react readily with most Organic Materials or Reducing Agents with very little energy that has to go into them.” – Trace Blackmore
“A Reducing Agent is a substance that can be oxidized by losing some of its electrons. An Oxidizing Agent is a substance that can be reduced by gaining electrons.” – Trace Blackmore
“We generally use Chlorine or Bromine, and we will commonly hear them referred to as either Oxidizers or Halogens.” – Trace Blackmore
“What Halogens are you using, and why are you using those?” – Trace Blackmore
“The OT Test (Orthotolidine) only tests for Total Halogen.” – Trace Blackmore
“The DPD Method (N, N-diethyl-p-phenylenediamine) can test for both Free and Total Halogen.” – Trace Blackmore
“Your Tests are your Tools. Don’t run your Test, and have your test tell you what to do next. Your Test only confirms or disproves any theory that you have.” – Trace Blackmore
“Diagnose first and then use a test to confirm.” – Trace Blackmore
“Free (Halogen) is the amount of Oxidizer in the system that hasn’t been combined with anything yet. It is Free to react; therefore Free is the amount of quick kill agent you have in your system to sanitize contaminants” – Trace Blackmore
“The Total Oxidizer is the amount of Free Oxidizer that you have plus the amount of Oxidizer that has combined with other Contaminants. So Free + Combined = Total” – Trace Blackmore
“Combined Oxidizer is the amount of Oxidizers that has combined with Organics.” – Trace Blackmore
“Make sure that whatever you are using is registered for that intended use.” – Trace Blackmore
Connect with Scaling UP! H2O:
Email: corrine@blackmore-enterprises.com (podcast producer)
Submit a show idea: Submit a Show Idea
Trace Blackmore on LinkedIn: in/traceblackmore/
Scaling UP! H2O on Facebook: @H2OScalingUP
Scaling UP! H2O on YouTube: ScalingUpH2o.com/YouTube
Links Mentioned:
When You Give, People Give Back – My episode on the Water We Talking About? Podcast
How to Apply or Renew Your Passport?
157 Pinks and Blues When Your Pinks Don’t Turn Pink
Events:
Stormwater Summit 2022 – June 27 to 29, 2022 in Minneapolis, Minnesota
2022 BOMA International Conference – June 25 to 28, 2022 in Nashville, Tennessee
The International Water Conference – November 6 to 10, 2022 in Orlando, Florida
Association of Water Technologies’ 2022 Annual Convention and Exposition – September 21 to 24, 2022 in Vancouver, Canada
How to Apply or Renew Your Passport?
Books Mentioned:


